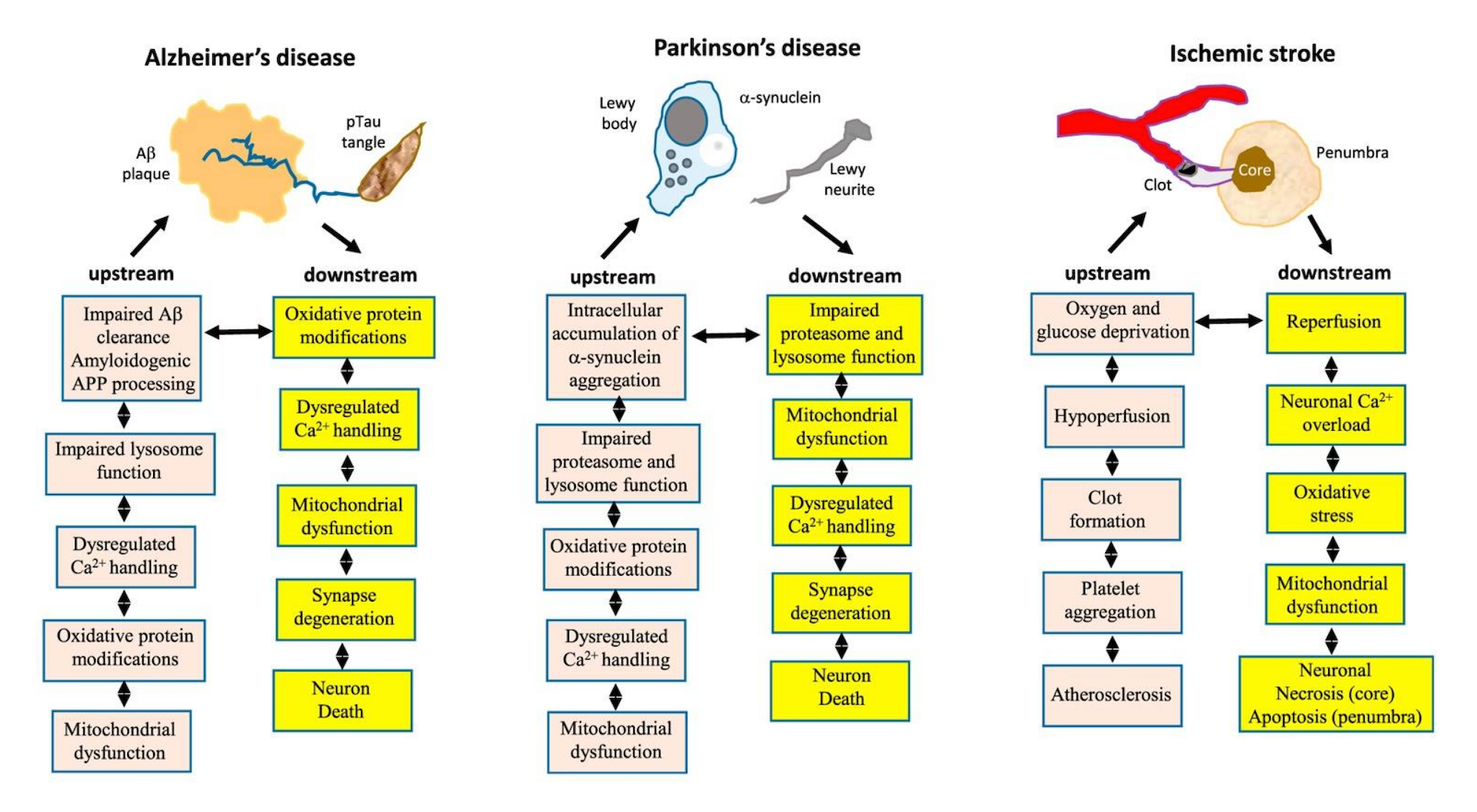
The six Braak stages describe the first appearance of cortical tau tangles in the transentorhinal cortex, where they spread throughout the medial and basal temporal lobes, then into nearby allocortex, next into isocortical associative regions, and finally into the unimodal sensory and motor cortex ( 17). Tau tangles are thought to exhibit a stereotypical pattern of cortical spread, which has been formalized into the Braak staging system ( 3, 17). 
A better understanding of tau pathophysiology is therefore of imminent need in order to aid development of these interventions. For these reasons and others, the focus of treatment discovery has shifted recently to tau, and numerous therapeutic interventions are currently undergoing research and development ( 16). Furthermore, cortical tau colocalizes with cortical atrophy and predicts future neurodegeneration ( 10), while the appearance of tau in specific cognitive networks leads to domain-specific cognitive impairments ( 15). However, the presence of A β is associated with the appearance of tau tangles outside of the MTL, which is itself highly associated with cognitive impairment ( 14). Tau tangles in the medial temporal lobe (MTL) are a very common age-related phenomenon ( 11), possibly associated with limited neurodegeneration and/or decrements in episodic memory ( 12, 13). However, the extent of A β burden is not as well correlated with cognitive status ( 7, 8) or local neurodegeneration ( 9 ?, 10) as is the extent of tau burden. Leading hypotheses have postulated these two hallmark proteins, A β and tau, either alone or in combination, are causative agents in disease etiology and progression ( 4- 6). 
At autopsy, AD presents with diffuse extracellular and neuritic β-amyloid (A β) plaques, and intracellular neurofibrillary tangles and neuropil threads of hyperphosphorylated tau, along with extensive neurodegeneration ( 3). Together, our results suggest variation in tau pathology is common and systematic, perhaps warranting a re-examination of the notion of “typical AD”, and a revisiting of tau pathological staging.Īlzheimer’s disease (AD) is the leading cause of dementia worldwide ( 1) and prevalence is expected to double in the next twenty years ( 2). Additionally, network diffusion models implicated that pathology originates and spreads through distinct corticolimbic in the different subtypes. Across all subtypes, younger age was related to worse cognition and more rapid tau accumulation. The subtypes presented with distinct demographic and cognitive profiles and differing longitudinal outcomes, however, no “typical” variant predominated. These “subtypes” were stable during longitudinal follow-up, and could be replicated in a separate sample using a different radiotracer. We replicated previously described limbic-predominant and medial temporal lobe-sparing variants, while also discovering posterior and lateral temporal subtypes resembling atypical clinical variants of AD. We identified four distinct trajectories of tau progression, ranging in prevalence from 18–33%, with no one progession predominating. 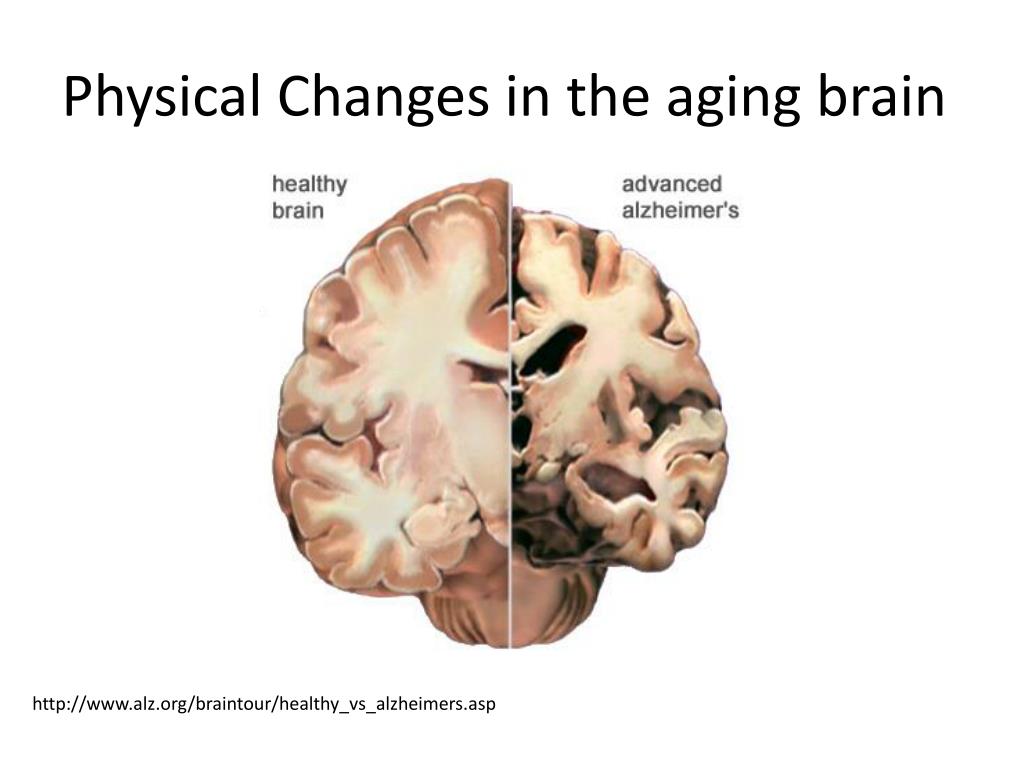
We analyzed 1612 tau-PET scans and applied to this sample a disease progression modeling framework designed to identify spatiotemporal trajectories of pathological progression. Still, a systematic, unbiased, wholebrain characterization of spatiotemporal variation in tau deposition in AD is lacking. The pattern of spread is thought to be fairly consistent across individuals, though more recent work has demonstrated substantial variability in the AD population that is often associated with distinct clinical phenotypes. Alzheimer’s disease (AD) is characterized by the progressive spread of tau pathology throughout the cerebral cortex.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
